Detection of Inflicted Bruises by Alternate Light: Results of a Randomized Controlled Trial
Katherine N. Scafide Ph.D., R.N.
Daniel J. Sheridan Ph.D., R.N.
Nancy R. Downing Ph.D.
R.N.,Matthew J. Hayat Ph.D.
Annotation
Since bruises are often difficult to detect on victims of violence, potentially impacting investigation and prosecution, this randomized controlled trial measured the effectiveness of an alternate light source (ALS) within visible and long ultraviolet spectrums at improving bruise detection compared to white light over time; and it also examined the effects of skin color, age, gender, localized fat, and injury mechanism on bruise detection.
Abstract
Bruises are often difficult to detect on victims of violence, potentially impacting investigation and prosecution. The purpose of our randomized controlled trial was to measure the effectiveness of an alternate light source (ALS) within visible and long ultraviolet spectrums at improving bruise detection compared to white light over time. We also examined the effects of skin color, age, gender, localized fat, and injury mechanism on bruise detection. Participants included 157 healthy adults with balanced sampling across six skin color categories. Bruises were created under the controlled application of a paintball pellet and dropped weight to one upper and lower arm, respectively. Using a crossover design, both bruises were examined 21 times over 4 weeks. Ten different wavelength (350–535 nm) and filter (yellow, orange, red) combinations were used. Multilevel models were used to analyze 2903 examinations on both upper and lower arms. Results in multivariable models showed after controlling for other covariates 415 and 450 nm using a yellow filter had greater odds of detecting evidence of bruising than white light (Upper Arm: 415 nm: OR = 5.34, 95% CI: 4.35–6.56; 450 nm: OR = 4.08, 95% CI: 3.36–4.96). Under either light source, being female and having more localized fat had increased odds of detecting bruises created by the dropped weight (female: OR = 2.96, 95% CI: 2.37–3.70; fat: OR = 1.21, 95% CI: 1.09–1.34). Our results support ALS as an appropriate tool to enhance concurrent physical assessment of bruises in the presence of known history of injury. Future development and evaluation of clinical practice guidelines for ALS application are needed.
Bruising is one of the most common types of soft tissue injury noted on victims of violence, including intimate partner violence, sexual assault, child abuse, and vulnerable adult abuse 1-4. Such injuries are usually caused by blunt, compressive or squeezing force trauma resulting in damaged blood vessels 5, 6. Evidence of bruising is usually identified by forensic clinicians through observed skin discoloration stemming from exsanguinated blood and its associated inflammation 7. Injuries, such as those from strangulation, can significantly impact clinical outcomes if they are not detected 8. Additionally, research indicates injury documentation is associated with greater victim engagement in the criminal justice process 9, 10 and may provide corroborative evidence in court 11.
Many factors contribute to being able to observe a bruise under normal lighting conditions, including the amount of extravasated blood 5; location on the body 12; depth 12, 13; subject’s age 14, 15; bruise’s age 15-17; and skin color 16. When blood is released into the extravascular space, hemoglobin is broken down through enzymatic processes to include bilirubin as one of its byproducts. On spectrophotometric analysis, hemoglobin exhibits the greatest amount of light absorption at a narrow peak of wavelengths around 415 nm, with a secondary, broader peak in light absorption around 543 and 576 nm 17, 18. Bilirubin has a broad absorption peak around 460 nm 18. Gross observation of light absorption by these molecules requires the use of alternate light, specific wavelengths which may include the visible (400–700 nm) or long ultraviolet (290–400 nm) spectrums 19. Generally, light is reflected, transmitted, scattered, and/or absorbed by the skin’s surface 20, 21 Transmitted light can be absorbed and reflected by deeper structures, with longer wavelengths generally having greater skin penetration 20. Filters (e.g., colored goggles and camera lenses) can then be used to block the reflected light allowing the absorbed light to appear darker by comparison 19.
An alternate light source (ALS) has been suggested by the U.S. Department of Justice as a tool to assist in identifying evidence of “subtle injury” 22. However, prior research on whether an ALS is effective at clinically detecting bruising within the visible and ultraviolet spectrums is limited 21, 23-25. The few available studies have found use of an ALS enhanced bruise observation, but researchers were unable to control for the potential effects of false positives either through study design 21, 24, 25 or execution 23. Confounding factors, such as pre-existing skin conditions and topical products, could mimic bruising by producing absorption under alternate light 21, 26, 27. Additionally, none of the research to date has examined how skin color impacts detection of bruising using an ALS. Given melanin is a major chromophore contributing to skin color, with an absorption spectrum overlapping that of hemoglobin 28, its effect on the ALS performance needs to be understood.
The aim of our study was to determine whether an ALS is more effective than white light at detecting bruises induced on diverse skin tones. Using an experimental design, we induced bruises using two different, published mechanisms, while controlling for factors affecting absorption detection. Eleven different bandwidth and filter combinations within the narrow band visible and ultraviolet spectrums were evaluated through repeated observations over 4 weeks. We hypothesized that wavelengths within the narrow absorption peak of hemoglobin would likely provide the greatest chance of detection.
See also the instructional video "Evidence Photography — Alternate Light Sources"
Methods
Study Design
A randomized controlled trial with a crossover design was used to address the study aim. Longitudinal data were collected over repeated participant visits. The order of light application (ALS or white light) was randomized for each bruise assessment to limit possible detection carryover effect.
Setting and Sample
The study was conducted at two large public universities in different regions of the United States. George Mason University (GMU) in Fairfax, VA and Texas A & M Health Sciences (TAM) in Bryan, TX both have large student populations from diverse backgrounds, providing the opportunity to recruit a convenience sample with a range of skin colors. Inclusion criteria for participation comprised of healthy adults aged 18–65. Exclusion criteria included use of medications and/or health conditions that affected coagulation and/or inflammation; history of prolonged or unusual healing; injuries, lesions, or artifacts visible under white light or ALS on possible bruise induction sites (left and right lateral deltoids, left and right anterior forearms); and upper arm circumference <24 cm.
Quota sampling was used to recruit an equal proportion of subjects across six skin color categories—very light, light, intermediate, tan, brown, and dark. Participants were excluded if quota was met for their given skin color. Skin color was measured on the right lateral deltoid with a spectrophotometer (Minolta® CM-600D; Konica Minolta, Osaka, Japan) using the Commission Internationale de l’Eclairage (CIE) L*a*b* color space 29. The average of three colorimetry readings was used. Category for skin color was determined by calculating the individual topology angle (ITA) using the L* (lightness) and b* (blue-yellow coloration) values in the following formula 30: ITA° = [tan-1((L*-50)÷b*)] × 180 ÷ p. Higher values indicate a lighter skin color, with the following cutoff angles for each category: very light > 55° = light > 41° = intermediate> 28° = tan > 10° = brown > -30° = dark 30. The spectrophotometer was calibrated as per manufacturer’s recommendations.
Sample size was determined with an a priori power analysis. The baseline area under a receiver operating characteristic curve (AUC = area under curve) was estimated, representing the probability of detecting bruises using white light based on research by Lombardi et al. 23. Assuming 80% power, a level of significance of 0.05, and detection of improved AUC of 10% between white light and ALS, a sample size of 130 subjects was needed. The target sample size was inflated to account for a conservative 20% attrition, resulting in 156 subjects. The planned large number of repeated observations on each subject increased statistical power to assess and quantify multivariable relationships.
Ethics
The study protocol was approved by the Institutional Review Boards at both institutions and conducted in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). Informed consent was obtained from all participants prior to screening for eligibility. A data safety monitoring board of independent experts provided ongoing review of data to address any participant safety concerns. Participants were compensated USD$260.
Screening Visit
Potential participants (N = 238) completed a comprehensive screening within 30 days of anticipated bruise induction (see flow diagram in online supplemental material). Both upper and lower arms were screened under white light and ALS for existing lesions or absorption artifacts. Self-reported gender and height were obtained. One lower arm and one upper arm were then randomly selected on each participant using a computerized random number generator (Microsoft Excel, 2015). Arm circumference (AC) of the target arms was measured at the approximate sites of the anticipated bruise inductions: halfway between the elbow and acromion process (upper arm) and 5 cm (2 in) distal to the elbow (lower arm). Using Lange Calipers (Beta Technology, Houston, TX), skinfold (SF) thickness was measured at the triceps and medial aspect of the forearm 5 cm from the elbow. Using the Heymsfield et al. 31 arm muscle area (AMA) equations (AMA = ([AC – SFp]2/4p) – x, where x is 10 for men and 6.5 for women), the Arm Fat Index (AFI), or proportion of fat, was computed as follows: AFI = [(AC2/4p – AMA)/AC2/4p]*100 31, 32. No such equation exists for the forearm. Finally, participants were weighed by digital scale (seca, Chino, CA). For safety purposes, participants were instructed to avoid medications that might increase risk of bleeding (e.g., ibuprofen and naproxen) for 72 h before and after bruise induction.
Bruise Induction
Prior to bruise induction, target arms were re-assessed under white light and ALS to assure no new artifacts or injuries were present since the screening visit. Bruise inductions (N = 164 participants) were performed by the researchers and trained research nurses using two different mechanisms. First, the lower arm bruises were induced by dropping a 6-oz ball bearing (Boca Bearing, Boynton Beach, FL) down a vertical, 1.5 m (5 ft) polyvinyl chloride (PVC) pipe, a method adapted from Lombardi et al. 23. Participants were positioned on a chair with the target arm placed horizontally on a table, palm-side up. The vertically secured PVC pipe was placed above the skin surface, 5 cm from the antecubital fossa and avoiding any visible vasculature. In order to identify the target area in future assessments, four permanent ink dots were placed on the arm approximately 6 cm from center of target using a transparent, rectangular template. The weight was then dropped on the lower arm and participants asked to rate their pain on impact using a 0 to 10 scale (0 = no pain; 10 = the worst pain imaginable).
Using a method developed by Scafide et al. 16, the second bruise was induced on the upper arm with a paint-filled projectile (paintball) fired at 6.1 m (20 feet) from a compressed air gun (Planet Eclipse GTEK, Manchester, UK). The gun was mounted on a stand and directed by a laser. Participants stood behind a full coverage, plywood barrier with their lateral aspect of their arm placed flush against a 5 by 10 cm cutout covered with two layers of 20 mil rubber. The paintball pellet was then discharged without warning (to prevent muscle tightening), and the participants were immediately asked to rate the pain of the impact. Participants were excluded if paintball did not directly impact skin or if abrasion occurred that prevented further assessment. The bruised area was marked in a same manner as the lower arm using the previously described template.
Bruise Assessments
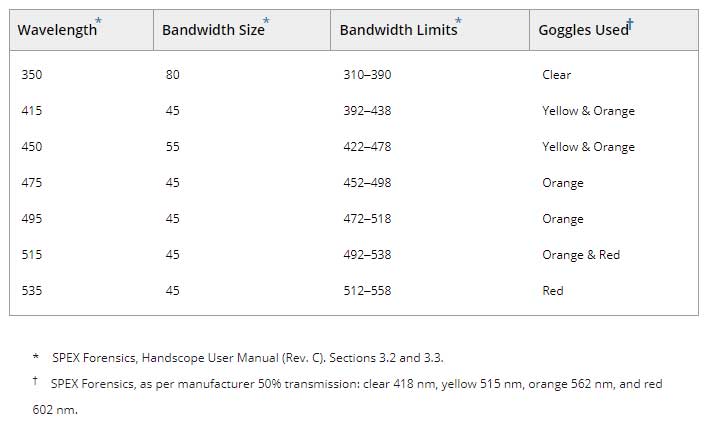
The target arm sites were assessed for evidence of bruising 21 times over 4 weeks postbruise induction on a set schedule (Figure 1 and online supplemental material). The first bruise assessment (Visit 1) occurred 30 min postbruise induction to allow time for the initial histamine response to subside 13. Bruise age at assessment was measured in hours since induction to accommodate variation in time between visits. The target arm sites were assessed for detection of bruising at each visit using two light sources: a dimmable 5600-Kelvin white light LED panel (SpectroLED Essential 240 Daylight; Genaray, New York, NY) and a multiwavelength alternate light device (Handscope® Xenon HSX-5000; Horiba, Piscataway, NJ). The order of the control and treatment light sources was randomized using an online data collection platform (Qualtrics, Provo, UT). The combinations of wavelengths and filters used during the assessment are presented in Table 1. Bruise detection was defined as the presence of any discoloration under the skin visible under white light or darkened area in contrast to the surrounding skin as viewed using the ALS at the point of trauma. Permanent ink dots were reapplied as necessary, and participants were queried whether their arms had been injured between visits.
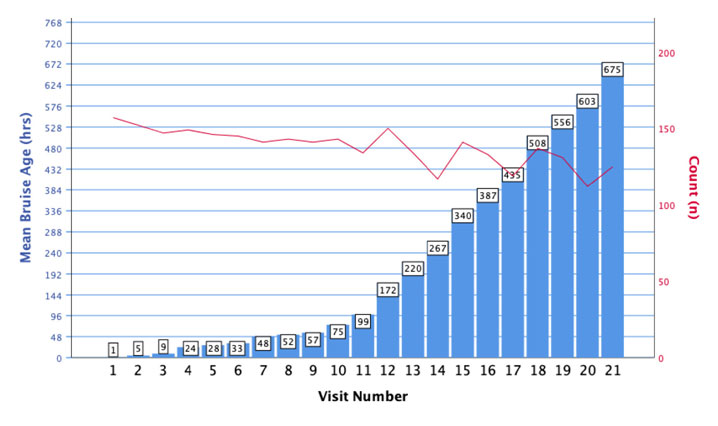
This dual axis graph depicts the mean bruise age in blue (left axis) and number of participants out of 157 who completed in red (right axis) at each scheduled bruise assessment visit. Visits 1–9 occurred three times-a-day over the first 3 days at least 4 h apart from the start time of assessment. Visits 10 and 11 were scheduled once-a-day the following 2 days followed by visits 13-21 occurring three times-per-week until 4 weeks postbruise induction.
Table 1. ALS wavelength and goggle color combination.
Fourteen researchers and trained research nurses conducted bruise assessments (6 GMU, 8 TAM). Observers were screened for corrected visual acuity of at least 20/30 (Snellen Chart) and color blindness (Ishihara Test). Inter-rater checks were conducted throughout the study to evaluate and support consistency of detection between observers (white light: Kappa 0.65; ALS: Kappa = 0.76).
Data Analysis
Descriptive statistics were used to summarize all study measures. The study data from this crossover randomized controlled trial entailed a complex multilevel structure. We anticipated 72,072 data points (312 bruises [156 upper arms, 156 lower arms] × 21 bruise assessment visits × 11 observations [white light and 10 ALS wavelengths]). Multiple assessments of each bruise were taken over time and at each visit resulting in multilevel and correlated data. Advanced statistical techniques using multilevel models were applied to account for this complex data structure. Marginal models with generalized estimating equations were used to model the dichotomous detection outcome. A three-level model was fit, with assessments nested in wavelength nested in bruise. Each model included a fixed effect for wavelength and any other covariates known based on theory to be associated with bruise detection. Skinfold and arm circumference were included as fixed effects in models for lower arm bruise and arm fat index for the upper arm bruise. A level of significance of 0.05 was used. The SAS Software System 9.4 (SAS Institute, Cary, NC) was used for statistical analyses.
Results
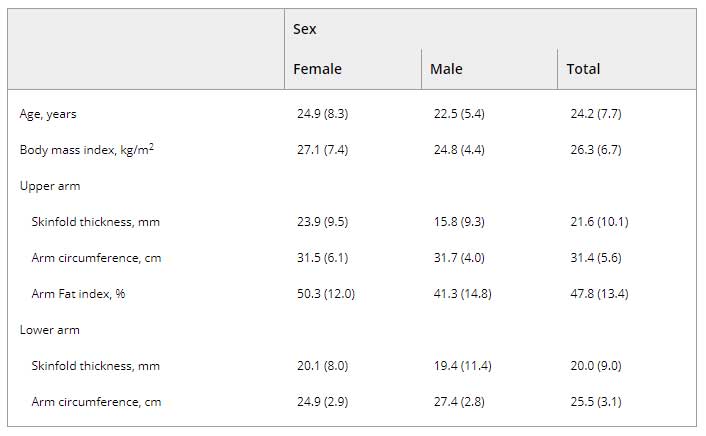
Between June 2017 and March 2019, 157 subjects participated in this study. Only 8 withdrew early prior to study completion, resulting in a 95% retention rate. Their data were retained for analysis. Recruitment was relatively equal between study sites (n = 81 at GMU, and n = 76 at TAM). The sample was mostly young (mean 23.9 years, SD 7.6) female (n = 114, 73%) and nearly equally distributed between all six skin color categories (Table 2). Skin color varied between and within reported race/ethnicity designations. Body composition characteristics are presented in Table 3. Most participants were right-handed (n = 141, 90%).
Table 2. Sample by skin color category (n = 157).
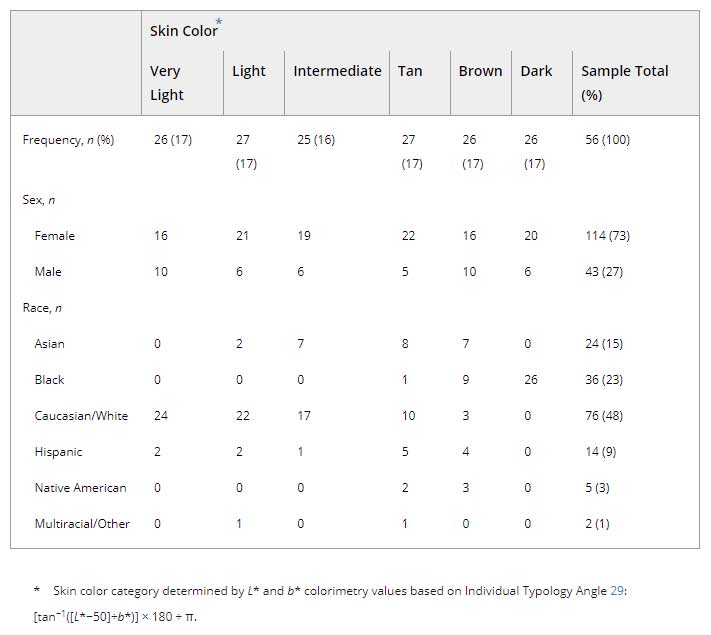
Table 3. Mean (SD) of body composition characteristics stratified by sex (n = 157).
Random arm selection for bruise induction resulted in equal number of left and right upper arms (left n = 79, 50.3%) and an oversampling of the left lower arm (n = 95, 60.5%). The 157 subjects were examined on average 19 times over the course of the experiment totaling in 2903 bruise assessment visits. At each visit, the upper arm and forearm bruised areas were assessed under white light and 10 ALS wavelength/filter combinations, providing a total of 31,621 observations on the upper arm and 31,509 on the lower arm. Participants rated pain of paintball impact on average 6.6 out of 10 (range 1–10). Bruises were visible on 100% of participants under white light at the first assessment (30 min postinduction). The dropped weight mechanism received a lower average impact pain score of 2.8 (range 0–7) with 86.6% (n = 136) of participants developing a bruise visible under white light during the study. A delay in first detection of some lower arm bruises by white light was noted; 20.5% (n = 28) of the injuries were first identified beyond the day of bruise induction.
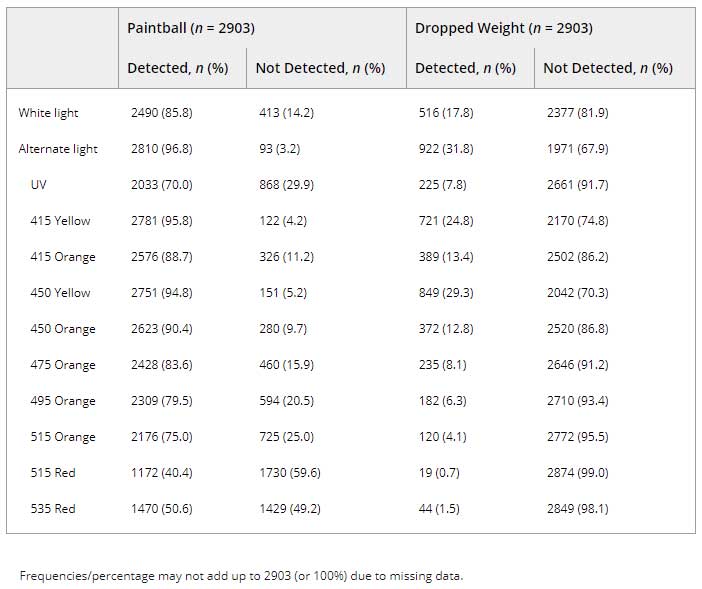
Table 4 shows the frequency of bruise detection over the total number of visits for all participants during the course of the study. Absorption was detected under alternate light more frequently than visible discoloration under white light for both upper and lower arms. Using the ALS, evidence of bruising was most frequently observed under 415 nm or 450 nm with the yellow filter (Figure 2). Of the 126 participants with recorded observations at 4 weeks postinduction, more bruises were visible on the upper arm under ALS (n = 103, 81.8%) than white light (n = 64, 50.8%).
Table 4. Frequency of bruise detection by light source and filter.

Examples of bruises on different skin colors observed 30 min (Visit 1) and 4 weeks (Visit 21) after bruise induction. Digital images taken using Canon T6i SLR with 50 mm fixed lens F2.2 ISO 400 with variable shutter speed and yellow filter (GG455; http://www.edmundoptics.com).
Results of multivariable marginal models are presented in Table 5. For both trauma mechanisms, alternate light wavelengths of 415 and 450 nm using a yellow filter had greater odds of detecting a bruise than white light while controlling for subject characteristics (upper arm: 415 nm yellow: OR = 5.34, 95% CI: 4.35–6.56; 450 nm yellow: OR = 4.08, 95% CI: 3.36–4.96). The orange filter was also effective at detecting absorption but only on the paintball induced bruises using 415 and 450 nm wavelengths (415 nm: OR = 1.42, 95% CI: 1.20–1.68; 450 nm: 1.77, 95% CI: 1.50–2.10). All other wavelength (i.e., UV and 475–535 nm) and filter combinations had lower odds of detecting bruises compared to white light while controlling for participant and bruise characteristics (Table 5).
Table 5. Multivariable multilevel model of bruise detection under alternate light compared to white light.
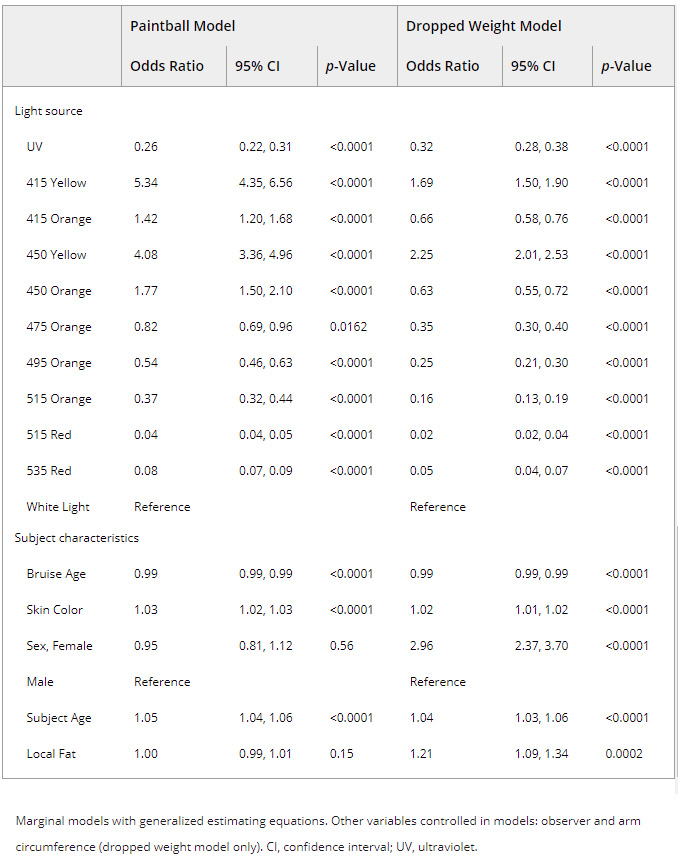
Multivariable modeling results also identified several subject factors affecting the likelihood of bruise detection regardless of light source used. Across both bruise mechanisms, controlling for other characteristics, the odds of detection were higher with increasing skin lightness (OR = 1.03, 95% CI: 1.02–1.03) and subject age (OR = 1.05, 95% CI: 1.04–1.06). Similarly, odds were almost 3 times greater for detecting the dropped weight bruises on women than on men (OR = 2.96, 95% CI: 2.37–3.70). Also, more localized fat contributed to increased odds of bruise detection using the dropped weight mechanism (OR = 1.21, 95% CI: 1.09–1.34). While controlling for other characteristics, the odds of detecting bruises created by either mechanism and viewed under any light source decreased by 12% for every additional 24 h postinjury (OR = 0.88, 95% CI: 0.88–0.89).
Discussion
For the last five years, forensic science practitioners of the National Institute of Justice’s Forensic Science Technology Working Group (TWG) have repeatedly requested to make detection of subtle injuries a research and development priority 33. Alternate light technology is already widely used in forensic science to identify latent or barely visible evidence at crime scenes and during victim examinations 19. However, research investigating its application in the clinical detection of bruising has been limited in both quantity and quality. Our study has advanced the science by attempting to use a more rigorous approach in both methodology and data analytics to better understand major factors associated with ALS identification of injuries on diverse skin tones.
As hypothesized, ALS wavelengths of 415 and 450 nm viewed through a yellow filter provided the best chances of bruise detection compared to white light. These results are consistent with the narrow absorption peak of hemoglobin, which fell within these bandwidths 17. The findings also support existing cadaver 34 and clinical research 23, 24, with only one exception. In their retrospective medical record review of 159 strangulation cases, Holbrook and Jackson 21 noted using an orange filter provided more frequent absorption detection than yellow. Their predominately African American sample (69%) may explain this phenomenon; theoretically, melanin’s broad absorption spectrum could be preventing any light reflection from being visible using the yellow filter 28. However, when we controlled for the effect of skin color in our analysis, the odds of detecting a bruise using the orange filter were less than yellow.
Not surprisingly, the ability to detect bruises under any light source diminished generally with time. However, we were able to establish through our repeated measures and modeling that regardless of bruise age in the first 4 weeks postinjury, 415 and 450 nm with a yellow filter outperformed white light in detecting evidence of bruising. Subsequent work should further investigate whether the age of bruises moderates the ability of specific ALS wavelengths to enhance visualization of these injuries. Our analysis is not designed to comment on the aging of bruises nor has other ALS research to date supported that possibility 35.
We examined several factors contributing to bruise detection under white and alternate light assessments. Because depth of bleeding could potentially alter the penetration and reflection of transmitted light, we chose to evaluate two different methods of bruise induction. Randeberg et al. 13 describe the use of paintball as a bruising mechanism being consistent with being struck by a whip given its high velocity and low mass. A dropped weight bruise mechanism was reported by Lombardi et al. 23 for the purpose of creating more subtle injuries with a heavier weight (4-oz in their case) and slower speed. Though we could not confirm this as fact, the bruise caused by the dropped weight mechanism should, theoretically, be deeper as a result of its likely longer contact duration. The delay in time for which the dropped weight bruises became visible on some participants suggests bleeding may not have been readily superficial.
The results of the two GEE models also differed—the odds of detecting bruises using the dropped weight method were greater with increasing localized fat or by being female. Subcutaneous fat, which is vascular, contributes in large part to the bleeding observed in bruise discoloration 15 as well as its size 16. Thus, our results may suggest the amount of fat could play a role in whether bleeding is visible for suspected deeper injuries to be detected. Alternatively, the force of the dropped weight may not have caused enough vessel damage on individuals with less subcutaneous fat to result in detectable bruising. The significance of sex as a factor may be associated with the higher fat distribution associated with being female.
Implications
The population of our study consisted of adults known to have experienced an injury to the area being assessed. In this context, information gathered during a proper medical-legal physical examination and interview can help support and/or eliminate possible alternative causes of light absorption. For example, we noted several instances of scars, tattoos, hyperpigmented areas, nevi, acne, and other lesions that demonstrated absorption under alternate light but distinguishable from bruising under direct white light assessment. In addition, washing the skin before ALS application, as we did, may remove topical products (i.e., cosmetics and sunscreen) known to cause light absorption 26, 27. Performing follow-up assessments in clinical practice may also capture changes in absorption over time more consistent with a healing bruise. However, in the absence of trauma-related history or other physical findings consistent with bruising, caution should be used when interpreting light absorption in isolation. Lombardi et al. noted low specificity of latent injuries on individuals providing no history of trauma 23.
Despite DOJ recommendations for the use of ALS 22, no evidence-based clinical practice guidelines exist for using ALS during injury assessment. Future development of standardized methods of alternate light application and documentation for this purpose must take into consideration the various stakeholders impacted (e.g., patients, clinicians, police and attorneys). ALS is not a diagnostic tool for bruising. Users must be sufficiently trained on both the science and techniques (particularly photography) to use the equipment safely and properly interpret its findings. Purchase of violet or blue ALS flashlights may be a cost-effective option for conducting bruise assessments. However, when selecting any ALS, other factors, such as lumens, should be considered, given the considerable variation available 36. More research on how variations in ALS devices could impact bruise assessment is needed. Finally, future studies should evaluate the impact of programmatic implementation of ALS on both clinical and criminal justice outcomes.
Limitations
Our study had several limitations. First, it was not feasible to blind observers to where the arm was injured given the bruises were not intended to be latent. To address this challenge, we used a randomized, crossover design to reduce the carryover effect of comparing one light source to the other. Additionally, we chose to measure bruise detection through direct assessments of the injury instead of through ALS photography to be more consistent with clinical skin assessment practices. Image analysis could have potentially provided more objective measures of detection; yet, analysis of our inter-rater concordance was good. Our bruise assessments were limited to observation without consideration of other clinically relevant findings (e.g., presence of induration and pain).
During bruise induction, the speed of the paintball and dropped weight were not controlled. However, the comparison of alternate light to white light occurred within the same bruise (i.e., each arm was being compared to itself) and, thus, should be less impacted by variations between bruises. Also, using different locations for the two bruise mechanisms may have confounded the comparison between models given the anatomical variations between the upper and lower arms. Our decision to model the two mechanisms separately was based on the substantially fewer visible bruises created using the dropped weight mechanism. Future use of this particular bruise induction method is not recommended given the inability to noninvasively confirm latent bruising. Finally, our sample was mostly young adults. Further research on an older population is needed to fully understand the effect of age on alternate light detection of bruising.
Conclusion
The need for better forensic techniques to identify and document subtle bruises, particularly on victims who are of color, is well established. Clinical application of alternate light for the purposes of bruise detection has been proposed given the light absorption properties of hemoglobin and its breakdown products. Our randomized controlled trial sought to examine the effectives of an ALS compared to white light on a diverse sample using a large data set of repeated measures. We determined alternate light wavelengths consistent with hemoglobin absorption, 415 and 450 nm viewed through a yellow filter, provided five times greater odds of detecting bruises than white light. Other factors such as sex, localized fat, age, injury mechanism, and bruise age all contributed to whether a bruise was detected by either light source. Our results support findings from previous studies while advancing the science through more rigorous design and analysis. ALS, used in conjunction with an appropriate physical assessment and history, may enhance forensic documentation of bruising in cases of reported injury. However, development and evaluation of evidence-based clinical practice guidelines for ALS implementation are needed.
Acknowledgments
We would like to thank the many participants who volunteered for this study and the graduate research assistants for their dedicated work in supporting this project. Specifically, we would like to acknowledge Dr. Bongani Sibandze, Dr. Ghareeb Bahari, Dr. Susan Senko, and Mona Mohammadifirouzeh for their assistance with participant recruitment and data collection. Additionally, we want to thank Dr. Yesser Sebeh for helping with data analysis. Authors would like to additionally thank Ms. Nesibe Kutahyalioglu who assisted with data collection and the creation of Figure 2. Finally, we would like to acknowledge Dr. Neil Langlois for his guidance during early project development and critical review of this manuscript.
Notes
- This project was supported by Award No. 2016-DN-BX-0147, awarded by the National Institute of Justice, Office of Justice Programs, U.S. Department of Justice.
- The opinions, findings, and conclusions or recommendations expressed in this publication are those of the authors and do not necessarily reflect those of the Department of Justice.
- Presented at the Annual Conference of the International Association of Forensic Nurses, September 11–14, 2019, in New Orleans, LA; and accepted for presentation at the 72nd Annual Scientific Meeting of the American Academy of Forensic Science, February 17–22, 2020, in Anaheim, CA.
References
- Kemp AM, Maguire SA, Nuttall D, Collins P, Dunstan F. Bruising in children who are assessed for suspected physical abuse. Arch Dis Child 2014; 99(2): 108– 13.
- Alempijevic D, Savic S, Pavlekic S, Jecmenica D. Severity of injuries among sexual assault victims. J Forensic Leg Med 2007; 14(5): 266– 9.
- Sheridan DJ, Nash KR. Acute injury patterns of intimate partner violence victims. Trauma Violence Abuse 2007; 8(3): 281– 9.
- Wiglesworth A, Austin R, Corona M, Schneider D, Liao S, Gibbs L, et al. Bruising as a marker of physical elder abuse. J Am Geriatr Soc 2009; 57(7): 1191– 6.
- Langlois NE. The science behind the quest to determine the age of bruises—a review of the English language literature. Forensic Sci Med Pathol 2007; 3(4): 241– 51.
- Ellis P. Cutaneous findings in children. In: KA Collins, RW Byard, editors. Forensic pathology of infancy and childhood. New York, NY: Springer, 2014; 243– 65.
- Ross C, Byard RW, Langlois NEI. Does the intensity of the inflammatory reaction in a bruise depend on its proximity to the site of trauma? Forensic Sci Med Pathol 2013; 9(3): 358– 62.
- De Boos J. Review article: non-fatal strangulation: hidden injuries, hidden risks. Emerg Med Australas 2019; 31(3): 302– 8.
- Fisher BS, Kaplan A, Budescu M, Fargo J, Tiller D, Everett J, et al. The influence of anogenital injury on women’s willingness to engage with the criminal justice process after rape. Violence Vict 2013; 28(6): 968– 83.
- Deutsch L, Resch K, Barber T, Zuckerman Y, Stone J, Cerulli C. Bruise documentation, race and barriers to seeking legal relief for intimate partner violence survivors: a retrospective qualitative study. J Fam Violence 2017; 32(8): 767– 73.
- Alderden M, Cross TP, Vlajnic M, Siller L. Prosecutors’ perspectives on biological evidence and injury evidence in sexual assault cases. J Interpers Violence 2018; 088626051877825. [Epub ahead of print]. https://doi.org/10.1177/0886260518778259
- Vanezis P. Interpreting bruises at necropsy. J Clin Pathol 2001; 54(5): 348– 55.
- Randeberg LL, Winnem AM, Langlois NE, Larsen ELP, Haaverstad R, Skallerud B, et al. Skin changes following minor trauma. Lasers Surg Med 2007; 39(5): 403– 13.
- Vanezis P. Bruising: concepts of ageing and interpretation. In: GN Rutty, editor. Essentials of autopsy practice. London, U.K.: Springer, 2001.
- Langlois NEI, Gresham GA. The ageing of bruises: a review and study of the color changes with time. Forensic Sci Int 1991; 50(2): 227– 38.
- Scafide KRN, Sheridan DJ, Campbell J, Deleon VB, Hayat MJ. Evaluating change in bruise colorimetry and the effect of subject characteristics over time. Forensic Sci Med Pathol 2013; 9(3): 367– 76.
- Hughes VK, Ellis PS, Burt T, Langlois NEI. The practical application of reflectance spectrophotometry for the demonstration of haemoglobin and its degradation in bruises. J Clin Pathol 2004; 57(4): 355– 9.
- Amazon K, Soloni F, Rywlin AM. Separation of bilirubin from hemoglobin by recording derivative spectrophotometry. Am J Clin Pathol 1981; 75(4): 519– 23.
- Marin N, Buszka JM. Alternate light source imaging: forensic photography techniques. London, U.K.: Routledge, 2013.
- Wright FD, Golden GS. The use of full spectrum digital photography for evidence collection and preservation in cases involving forensic odontology. Forensic Sci Int 2010; 201(1–3): 59– 67.
- Holbrook DS, Jackson MC. Use of an alternative light source to assess strangulation victims. J Forensic Nurs 2013; 9(3): 140– 5.
- U.S. Department of Justice. A national protocol for sexual assault medical forensic examinations: adults/adolescents, 2nd edn. Washington, DC: U.S. Department of Justice, Office on Violence Against Women, 2013; Report No.: NCJ 228119.
- Lombardi M, Canter J, Patrick PA, Altman R. Is fluorescence under an alternate light source sufficient to accurately diagnose subclinical bruising? J Forensic Sci 2015; 60(2): 444– 9.
- Mimasaka S, Oshima T, Ohtani M. Visualization of old bruises in children: use of violet light to record long-term bruises. Forensic Sci Int 2018; 282: 74– 8.
- Scafide KN, Sharma S, Tripp NE, Hayat MJ. Bruise detection and visibility under alternate light during the first three days post-trauma. J Forensic Leg Med 2020; 69: 101893.
- Pollitt EN, Anderson JC, Scafide KN, Holbrook D, D’Silva G, Sheridan DJ. Alternate light source findings of common topical products. J Forensic Nurs 2016; 12(3): 97– 103.
- Olds K, Byard RW, Winskog C, Langlois NEI. Validation of ultraviolet, infrared, and narrow band light alternate light sources for detection of bruises in a pigskin model. Forensic Sci Med Pathol 2016; 12(4): 435– 43.
- Shriver MD, Parra EJ. Comparison of narrow-band reflectance spectroscopy and tristimulus colorimetry for measurements of skin and hair color in persons of different biological ancestry. Am J Phys Anthropol 2000; 112(1): 17– 27.
- Commission Internationale d’Eclairage. CIE Colorimetetry. Part 4: 1976 L*a*b* colour space. Vienna, Austria: CIE, 2007; Report No.: CIE S 014–4/E:2007.
- Chardon A, Cretois I, Hourseau C. Skin colour typology and suntanning pathways. Int J Cosmet Sci 1991; 13(4): 191– 208.
- Heymsfield SB, McManus C, Smith J, Stevens V, Nixon DW. Anthropometric measurement of muscle mass: revised equations for calculating bone-free arm muscle area. Am J Clin Nutr 1982; 36(4): 680– 90.
- Frisancho AR. Anthropometric standards for the assessment of growth and nutritional status. Ann Arbor, MI: University of Michigan Press, 1990.
- National Institute of Justice, US Department of Justice. Forensic Science Research and Development Technology Working Group: operational requirements. https://nij.ojp.gov/topics/articles/forensic-science-research-and-development-technology-working-group-operational (accessed December 1, 2019).
- Olds K, Byard RW, Winskog C, Langlois NEI. Validation of alternate light sources for detection of bruises in non-embalmed and embalmed cadavers. Forensic Sci Med Pathol 2017; 13(1): 28– 33.
- Hughes VK, Ellis PS, Langlois NEI. Alternative light source (polilight®) illumination with digital image analysis does not assist in determining the age of bruises. Forensic Sci Int 2006; 158(2–3): 104– 7.
- Forensic Technology Center of Excellence. Landscape study of alternate light sources. Washington, DC: U.S. Department of Justice, National Institute of Justice, Office of Investigative and Forensic Sciences, 2018; Report No.: 251492.
 Detection of Inflicted Bruises by Alternate Light: Results of a Randomized Controlled Trial. Copyright for this article is retained by the authors. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License which permits unrestricted noncommercial use, distribution, and reproduction, provided the original work is properly cited and not changed in any way. Based on a work at https://nij.ojp.gov/library/publications/detection-inflicted-bruises-alternate-light-results-randomized-controlled.
Detection of Inflicted Bruises by Alternate Light: Results of a Randomized Controlled Trial. Copyright for this article is retained by the authors. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License which permits unrestricted noncommercial use, distribution, and reproduction, provided the original work is properly cited and not changed in any way. Based on a work at https://nij.ojp.gov/library/publications/detection-inflicted-bruises-alternate-light-results-randomized-controlled.
Article posted January 10, 2022

